Products
- Card Edge Connectors
- RECTANGULAR CONNECTORS
- D-Sub Connectors
- Machined D-Sub Connectors
- Power Combo D-Sub Connectors
- Parallel Port D-Sub Connectors
- Waterproof D-Sub Connectors
- DB9 Connector
- DB15 Connector
- DB25 Connector
- Vertical PCB D-Sub Connector
- D-Sub Panel Cut-outs
- Cable Wire D-Sub Connector
- PCB D-Sub Connector
- Right-angle PCB D-Sub Connector
- Backshells or Hoods, and Caps for D-Sub Connectors
- D-Sub Connector Housings
- D-Sub Connector Contacts
- Modular & Magnetic Jacks
- USB Connectors
- HDMI Connectors
- INLINE CONNECTORS
- HEADER CONNECTORS
- Spring Loaded Connectors
- Waterproof Connectors
- Cable Assemblies
- Custom Connectors
Engineering Considerations for Medical Connectors
The medical device industry is highly regulated and products are cleared by the FDA before they can even be marketed. This process categorizes these devices into different classes (I, II, and III) and, depending upon the risk to the patient and operator, will either require more or less oversight and control via standards compliance. Along with the medical devices and equipment, specific types of connectors are often subjected to the same level of scrutiny. These connectors will require strict compliance with the IEC 60601-1 standards to prevent harm to either the patient or operator. Connectors that are frequently handled by medical personnel, for example, will generally require polarization of the connector head as well as the ability to visually differentiate between connectors to prevent mismatches and possible disastrous consequences.
While this will be touched upon in this piece, this article will focus more on the connectors used within medical equipment (i.e., board-to-board, wire-to-board, and wire-to-wire) as well as ruggedized implementations of universally used connectors (e.g., USB, HDMI, D-sub). These connectors are not application-specific; however, they are still important to the functionality of medical equipment/devices and must often still be ruggedized to prevent them from malfunctioning within and outside of their electronic enclosure.
Medical device trends
Medical technology has evolved with the proliferation of wireless networks of all types (e.g., WAN, LPWAN, broadband, massive machine-type communications). Wireless body area networks (WBANs), for example, are intended to collect physiological data from sensors installed on a patient and wirelessly transmit the information for monitoring and tracking purposes. The growth of wearable devices has led to innovations in sensing, materials, and wireless data/power transfer, shrinking package sizes. Many medical applications will use advanced technologies to meet next-generation healthcare needs. The growth of AI has also spread into niche medical applications for diagnosis and even for surgery performed remotely with robotic arms (telesurgery). This trend calls for power-hungry processing tasks at the edge where power density and processing capability in increasingly small form factors are necessary. For connectors, this translates to higher pin counts and power ratings. However, these advances must not detract from their safety as this is the ultimate requirement for medical applications.
Medical device standards
The ISO 60601-1 standard is one of the basic standards used for medical equipment and specifies means of patient protection (MOPP) and means of operator protection (MOOP) for devices touching a patient or operator. These protection ratings specify creepage and clearance distances that most commercial connectors cannot meet where clearance is the shortest distance through air between two conductive parts, such as pins. Creepage is the shortest distance between two pins along the insulating material between them. These distances are meant to ensure that no arcing can occur between pins, causing potential harm to patients and handlers. The standard also specifies a maximum leakage current that flows from the probe contact to ground through the patient for three classifications: Type Body (B), Body Floating (BF), and Cardiac Floating (CF) as shown in Table 1.
IEC 60601-1 Medical Device Categories
Classification |
Applicable Areas |
Use Cases |
|---|---|---|
|
Type B |
Are not in direct contact with the patient’s heart |
Hospital beds, surgical lights, infusion pumps |
|
Type BF |
In direct contact with the patient but not with the patient’s heart |
Endoscopes, ultrasound equipment, transesophageal echocardiogram (TEE) probe |
|
Type CF |
In direct contact with the patient’s heart |
Heart monitors, electrocardiogram (ECG) machines |
Table 1: Medical-device categories, according to IEC 60601-1.
Some of the general standards for the different types of connectors and electronics found in medical equipment can be seen in Table 2.
Medical Application Standard Overview
Standard |
Description |
|---|---|
|
ISO 60601-1 |
Basic safety and essential performance requirements for devices and components that qualify as medical electrical equipment. |
|
ISO 5356 |
Standard for anesthetic and respiratory equipment (i.e., breathing systems, patient connectors, and common gas outlets), focusing on dimensional engaging requirements for the conical connectors used in these devices. |
|
ISO 80369 |
Standard that specifies requirements around patient safety by reducing the risk of misconnections between medical devices and accessories in enteral and gastric applications, neuraxial applications, intravascular, or hypodermic applications. |
|
ISO 13485 |
Outlines the requirements for a quality management system (QMS) specific to the medical devices industry. |
|
ISO 62304 |
Medical device software and software lifecycle processes. |
Table 2: An overview of various standards within medical applications.
As shown above, there is a great deal of regulation around electronic medical equipment from quality assurance and software to restrictions in medical device hardware realization. Designers and manufacturers must comply with these various labeling, quality assessment, materials, and other requirements to clear their products with the FDA. This is especially true when the equipment under question connects to a patient. There are, however, electronic devices and equipment that do not necessarily interact directly with a patient. These devices still require a degree of ruggedization against the potential stressors and strains of the healthcare environment.
Use case: Card edge, header, and in-line connectors in a blood analyzer
Medical equipment will use different types of modalities to test, monitor, or achieve a specific task. Many of these machines will use a mix of mechanical, hydraulic, pneumatic, electronic, optical, and radiological principles for imaging, sensing, actuation, and control.
A blood analyzer, for example, will count and identify blood cells at high speed with accuracy to determine a patient’s general health status and has become a routine analysis for various means of detection. The machine itself will take a sample, often manually drawn by venipuncture, and will prep the sample through dilution or centrifugation (i.e., spinning blood via motor) and use optical or electronic sensors for measurement. The sample may be broken down into smaller volumes (aliquoting) to perform multiple tests on a single sample. These aliquoted samples are then transported to various parts of the machine and combined with various reagents in a mixing chamber to perform these different tests with the use of hydraulic lines and valves. Samples can be dispensed via hydraulic dispensers into an analyzer cassette. A basic block diagram of a blood analyzer can be seen in Figure 1.
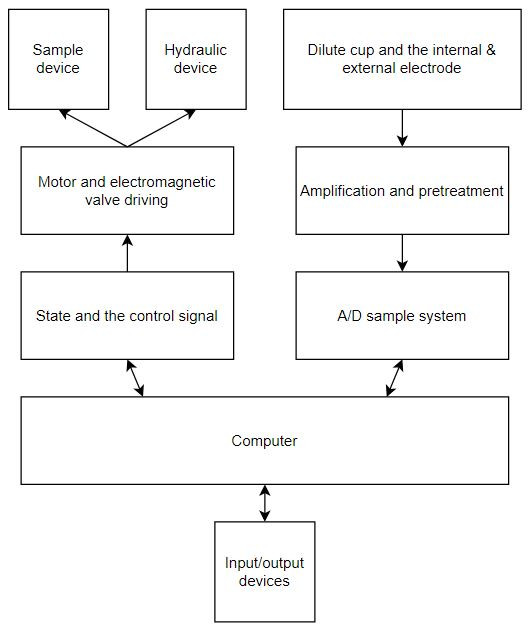
Figure 1: Basic block diagram of blood analyzer [1].
As shown above, this machine is quite sophisticated and will require precise timing and fluid controls to process many samples. The control board within the machine will use both headers and card edge connectors for signal and power delivery between PCBs. For these connectors to function reliably, they will require vibration resistance due to the use of mechanical methods such as centrifugation and hydraulic control systems. A degree of moisture and corrosion resistance may also be necessary due to the potential splashes from reagents.
Card edge connectors in medical equipment
Card edge connectors allow for PCBs with a series of pads along their edge to be inserted directly in the connector to interface with another system. These are often used in computers as expansion cards, memory modules, or data acquisition (DAQ) cards to connect to a high-speed interface such as PCIe. A blood analyzer might, for example, use DAQ cards in the A/D sample system to collect and process incoming sensor data. Or a patient-monitoring system could require card edge connectors to expand its storage or test capabilities.
EDAC has a massive range of card edge connectors for these types of applications. Higher gold-plating thicknesses on contacts ensure that the more reactive base metal used in the pins does not oxidize and negatively impact conductivity. Connectors in high-vibration environments are susceptible to fretting corrosion where the rubbing that occurs at the metallic joints of a connection can rub off the plating as well as the underlying material causing corrosion ― a condition that worsens with time. This type of abrasion also occurs with frequent mating cycles — not to mention the potential of some of these connectors to get splashed with reagents. A thicker plating will mitigate the risk of that potential failure mode.
Another risk that comes with vibration, especially in card edge connectors, is a potentially worsening connection or intermittent connections. Card edge connectors provide some elastic support in the event a circuit card is moving due to vibrations or transient shock. EDAC’s bellow-style contacts provide a good contact spring force to increase vibration resistance. After mating, a high-integrity connection is formed with the copper alloy contacts that have a low resistance of at most 10 mΩ (Figure 2).
Figure 2: Qualities of EDAC’s card edge connectors that make these connectors vibration and corrosion-resistant for medical equipment such as blood analyzers.
For additional vibration resistance, EDAC offers metal-to-metal card edge connectors with a high-reliability interlock mate. This mate uses an innovative hermaphroditic contact design. These identical contacts utilize a fork-like shape that slides together for a gas-tight mate. At the cross-section, the beveled edges of the contact pins float in the insulator, allowing for movement while maintaining a strong contact despite vibration and mechanical shock (Figure 3). This mate removes the rigidity associated with traditional card connections, where stiff leads could yield a shorter life than these interlocked types of mates.
Figure 3: EDAC’s card edge connector with a hermaphroditic contact design (left) and a cross-sectional view of the mate showing a gas-tight connection that can withstand movement due to the vibration and shock.
Header connectors in multi-board solutions for medical devices
Header connectors are universally used in all multi-board solutions to easily connect between boards. Typically, flexible cables such as ribbon cables are used as the cabling while headers are the connector heads. These connectors are inevitably found in control boards going into blood analyzers as well as other medical equipment such as X-rays, CAT scanners, ultrasound equipment, diagnostic cardiology equipment, MRI machines, heart-lung support systems, patient-monitoring systems, and more. They are the go-to cost-effective solution for wire-to-board and board-to-board connectors. EDAC offers standard box headers as well as more specialized E-Socket and E-Pin headers, high-power headers, and DIN headers. All of these may be suitable for medical applications.
The E-Socket receptacle and E-Pin male headers mate to provide reliable board-to-board connections. As shown in Figure 4, the fork design of the E-Socket receptacle can sufficiently grasp the E-Pin male connector with a maximum of 10 mΩ contact resistance. The high-conductivity copper alloy used in the pin connections offers both a low resistance and a better contact spring force. Up to 50µ'' of gold plating can be used for high resistance to corrosion and pin damage due to vibration. While a base material of UL 94V-0 Thermoplastic Polyester is used for an operating temperature range from -40o C to 105o C, other high-temperature materials can also be utilized for an extended temperature range. Given the durability of these connections, they can withstand a minimum of 200 mating cycles.
Figure 4: E-Socket and E-Pin headers will mate to form a vibration-resistant board-to-board connection.
For power connections between circuit boards, high-power headers can also be used — offering up to 20 amps of rated current with integrated ground plane contacts that are offset from the power connections. The power pins themselves have a sizeable pitch of 4 mm to mitigate the risk of arcing between contacts, thereby increasing the safety of the connection (Figure 5).
Figure 5: Power header connectors offer up to 20 amps of current with dual-row configuration and a 4mm pitch.
In-line connectors in medical equipment
Within medical equipment such as the blood analyzer use case listed above, the need for a wire-to-wire and wire-to-board solution such as an in-line connector may be necessary where a card edge or header connector may not be sufficient. In-line connectors are characterized by their relatively small pin count. These are ideal for wire harnesses within medical systems. In lab environments, in-line connectors may face exposure to fluids, potentially causing both corrosion and moisture ingress. This can lead to an intermittent or failed connection. EDAC’s in-line connectors are IP67-rated, making them completely dust-tight and able to withstand temporary immersion in water (Figure 6). EDAC accomplishes a waterproof connection with its E-Seal technology, which uses a proprietary epoxy sealing process to seal the entire back of the connector rather than the individual pins. This yields a complete seal with a smooth finish and guarantees no water ingress, as fully tested to IP67 standards.
Figure 6: EDAC’s in-line connectors can withstand immersion in water, making them resistant to splashes in a lab environment.
D-sub and HDMI connectors in medical equipment
D-subminiature connectors are popular interfaces for serial communications such as RS-232 or RS-485. The connector can be found in applications spanning from personal computers for residences to controllers for industrial automation. Medical devices and equipment will inherently require the use of sensors and actuators to monitor different vitals, measurements, and perform specific tasks. Figure 7 shows a D-sub connector that connects a defibrillator to measure oxygen saturation.
Figure 7: Philips D-sub-to-pulse-oximeter probe for a defibrillator. Source: Philips
A processing unit such as a DAQ system can then digitize any sensor signals (e.g., sensory data from blood analysis) and convert them to stream transmission protocols such as RS-232 via D-sub connectors to be analyzed by a local computer. DAQ systems are used for processing in many different medical use cases including imaging such as magnetic resonance imaging (MRI), positron emission tomography (PET), ultrasound, and diagnostics applications that demand higher data-acquisition bandwidths to both increase imaging resolution and allow for quicker results processing (Figure 8).
Figure 8: Plexon OmniPlex Neural Data Acquisition System with up to 512 neural recording channels. Source: Plexon Inc.
D-sub connectors and HDMI connectors can also be used to feed video signals to computers and monitors to view images. Point-of-care ultrasound (POCUS), for example, will use several video output options including VGA, DVI, and HDMI, as well as more legacy video outputs such as S-video or C-video (Figure 9). VGA, DVI, and VESA are variants of the D-sub connector optimized for video applications. EDAC offers all of these variations on the D-sub connector.
Figure 9: VGA, HDMI, and Ethernet ports seen on the Philips CX50 Ultrasound Machine. Source: Philips
D-sub connector engineering considerations
The various ruggedization methods for the header and card edge connectors also apply to D-sub connectors. These connectors can undergo constant vibration that will deteriorate the quality of the connection over time. Sufficient gold plating and proper mounting can help mitigate these effects. D-sub connectors within medical applications should be IP-rated to prevent damage from dust and moisture. EDAC offers D-sub connectors that are fully submersible with an IP67 rating that use the E-Seal technology described earlier. The connectors can be plated with up to 30µ” of gold to offer corrosion, vibration, and mechanical shock resistance.
Another consideration particularly for D-sub connectors in medical applications is the potential for both radiated and conducted emissions. Two of the major causes of equipment failing to meet emission requirements are connectors and cabling. Cable assemblies can act as an antenna, radiating unwanted noise from medical equipment. This noise can, for example, emanate from the machine’s internal power supply with high-speed switching devices. The cable can also conduct noise from other sources that it is in contact with and bring that to sensitive internal circuitry which can cause equipment malfunctions. EDAC’s D-sub connectors come with metal shells, when connected to chassis ground and to the shielding of a cable, this can effectively mitigate EMI (Figure 10).
Figure 10: IP67-rated D-sub cable assembly with a waterproof hood and rubber seal. Metal hoods can also be used where EMI protection is critical.
HDMI connector engineering considerations
HDMI connectors are a standard interface for displays. However, the conventional HDMI connector will likely fail or disconnect in a medical environment where vibration, mechanical shock, or moisture ingress may occur. EDAC’s waterproof HDMI connectors use the E-Seal technology for resistance to splashes and potentially corrosive substances. To offer a reliable, vibration-resistant connection, the connectors feature quarter-turn twist-and-lock over mold (Figure 11).
Figure 11: IP67-rated HDMI connectors with a quarter-turn twist-and-lock over mold to prevent disconnects in the event of vibration and shock.
Spring-loaded connectors for medical equipment
Connector solutions for wearables charging or production tests would need an easy blind mate with high durability. Spring-loaded (or pogo-pin) connectors can be adapted to fit medical applications such as hearing aids for an easy clip/unclip charging ability.
EDAC’s spring-loaded connectors are more convenient than standard pogo pins by using magnets to both provide polarization and increase the reliability of the connection (Figure 12). These connectors can maintain a connection in environments with vibration with a precision machined finish. One factor that maintains a high vibration resistance is a spring-loaded connector’s stroke distance. The stroke distance is the distance from the resting position of the pogo pin to its maximum compression position when mated. The stroke distance of EDAC’s pogo pins ensures that the connector can maintain reliable contact during vibration and during minor misalignments that would normally break a connection with standard contacts. With a durability of up to 30,000 mating cycles, the EDAC connectors can withstand the strains of frequent connectors and disconnects. The connectors are assembled into a high-temperature thermoplastic for a wide operating temperature range.
Figure 12: EDAC’s spring-loaded connectors can be adapted for testing/charging applications for wearables and medical device production.
Conclusion
Connectors used within medical equipment are often subject to stringent medical standards. This is particularly the case when the connector is directly connected to a patient or managed by medical personnel. While there are some application-specific connectors (e.g., a connector that mates to ultrasound equipment from the probe), the medical industry also makes use of connectors that are ubiquitously used in most electronic equipment. This includes wire-to-wire (in-line), wire-to-board (in-line, header), board-to-board (header, card edge) connectors, and other conventional connectors such as D-subs or HDMI. These connectors may need to be ruggedized against vibrations, and mechanical shock from handling (e.g., drops, crush), and will likely need to be both dust and watertight to mitigate the risk of failure from exposure to fluids and chemicals. EDAC offers a massive range of connectors for medical applications that meet these requirements.
References
- L. Tao, L. Luo and F. Li, "The design and implementation of blood analyzer control system based on the fluid flow aperture device," 2015 IEEE International Conference on Mechatronics and Automation (ICMA), Beijing, China, 2015, pp. 174-178, doi: 10.1109/ICMA.2015.7237477.
- Balter ML, Chen AI, Fromholtz A, Gorshkov A, Maguire TJ, Yarmush ML. System Design and Development of a Robotic Device for Automated Venipuncture and Diagnostic Blood Cell Analysis. Rep U S. 2016 Oct;2016:514-520. doi: 10.1109/IROS.2016.7759102. Epub 2016 Dec 1. PMID: 28239509; PMCID: PMC5321615.
Go Back











